
Making accuracy more affordable
Introducing ALLYcgm

Use of CGM (versus SMBG) has been associated with a 25.3mmol/mol decrease in HbA1c over 6-months¹
Proportion meeting the ADA HbA1c target of <53mmol/mol (<7%) at baseline and 6-month follow up¹
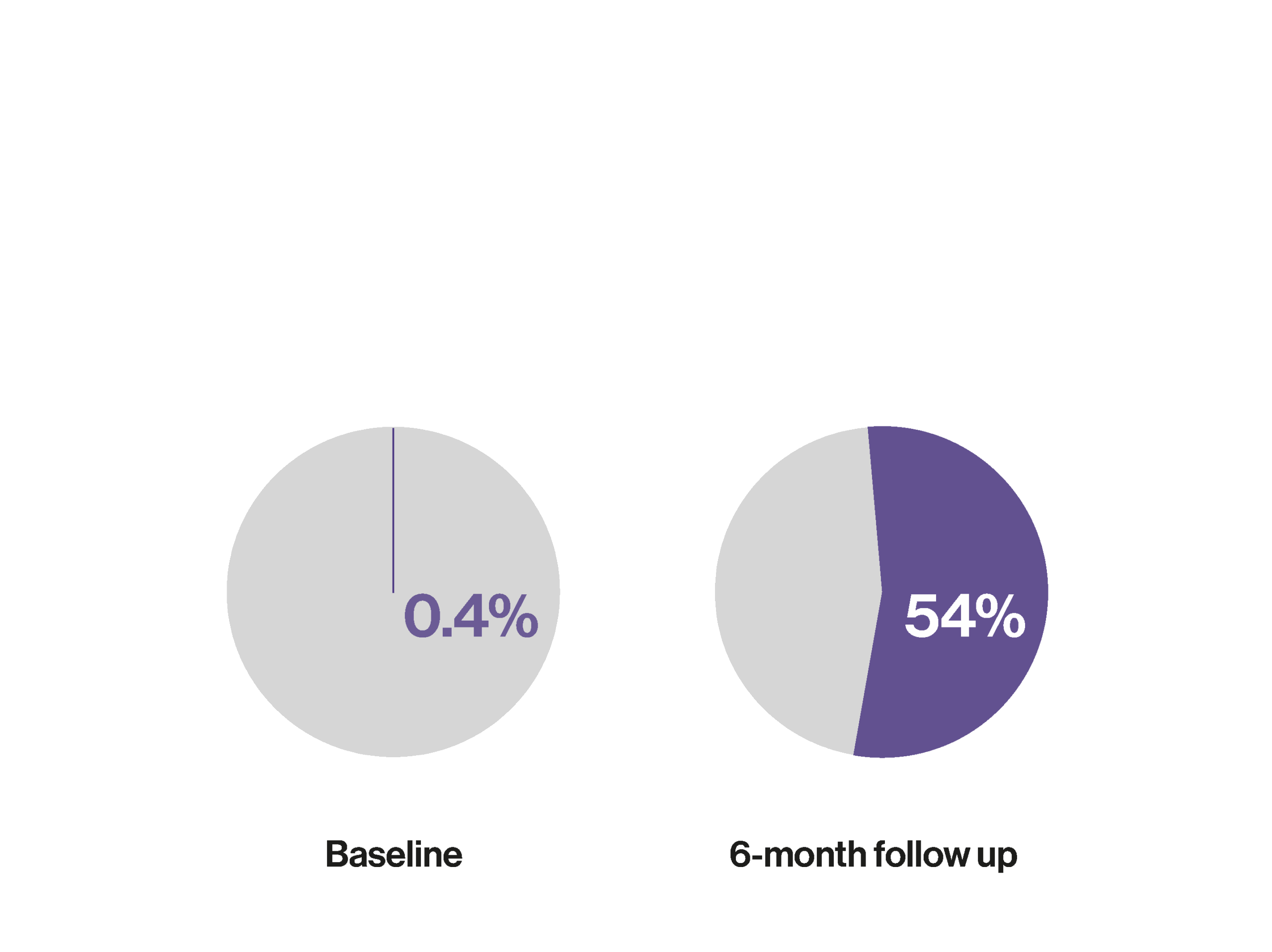
Results obtained with Dexcom G6. 1. Grace TP et al. Clin Diabetes. 2024 Aug 9;42(4):540-546.
Psychosocial benefits
Participants in a study credited CGM use with¹:
Making healthier lifestyle choices
Understanding how their diet
affected their glucose
Increased likelihood to walk or exercise in response to rising glucose levels
1. Ehrhardt N, Al Zaghal E. Clin Diabetes. 2020 Apr;38(2):126-131.
Nice guidance to prescribers
When choosing a continuous glucose monitoring (CGM) device¹:
Use shared decision making to identify the person’s needs and preferences, and offer them an appropriate device.
If multiple devices meet their needs and preferences, offer the device with the lowest cost.

offers:
Exceptional accuracy²
Class-leading features³
Lower cost to NHS than most reimbursed CGMs*

*Based on a comparison of the ALLYcgm drug tariff submitted price and the published prices of currently listed CGMs. References: 1. NICE Guidance NG17 1.6.11 updated August 2022. 2. Jendrike N et al, Journal of Diabetes Science and Technology 2025; 1–8. 3. 8100-10657 Rev B ALLYcgm User Manual.
Patients suitable for ALLYcgm²,³.
Estimate of the proportion of current CGM patients who could benefit from ALLYcgm once HCL and paediatric patients are excluded
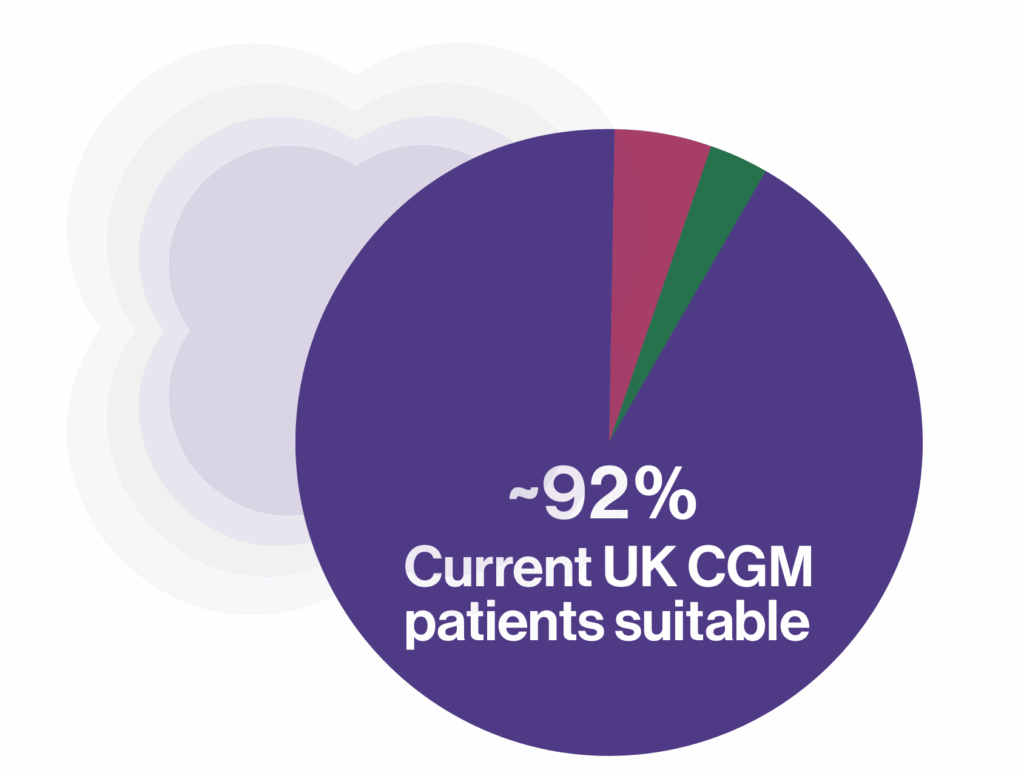
~5%
HCL users (not indicated)
~3%
Pediatric (not indicated)
Patients unsuitable³ for ALLYcgm
Patients under the age of 18
Patients using CGM as part of hybrid closed loop system
Pregnant and breastfeeding women
Dialysis patients and critically ill patients
HCL = Hybrid Closed Loop. 1. The UK insulin pump audit 2016-17. 2. DUK Position Statement. 3. 8100-10657 Rev B ALLYcgm User Manual.
Mobile Device sold separately.
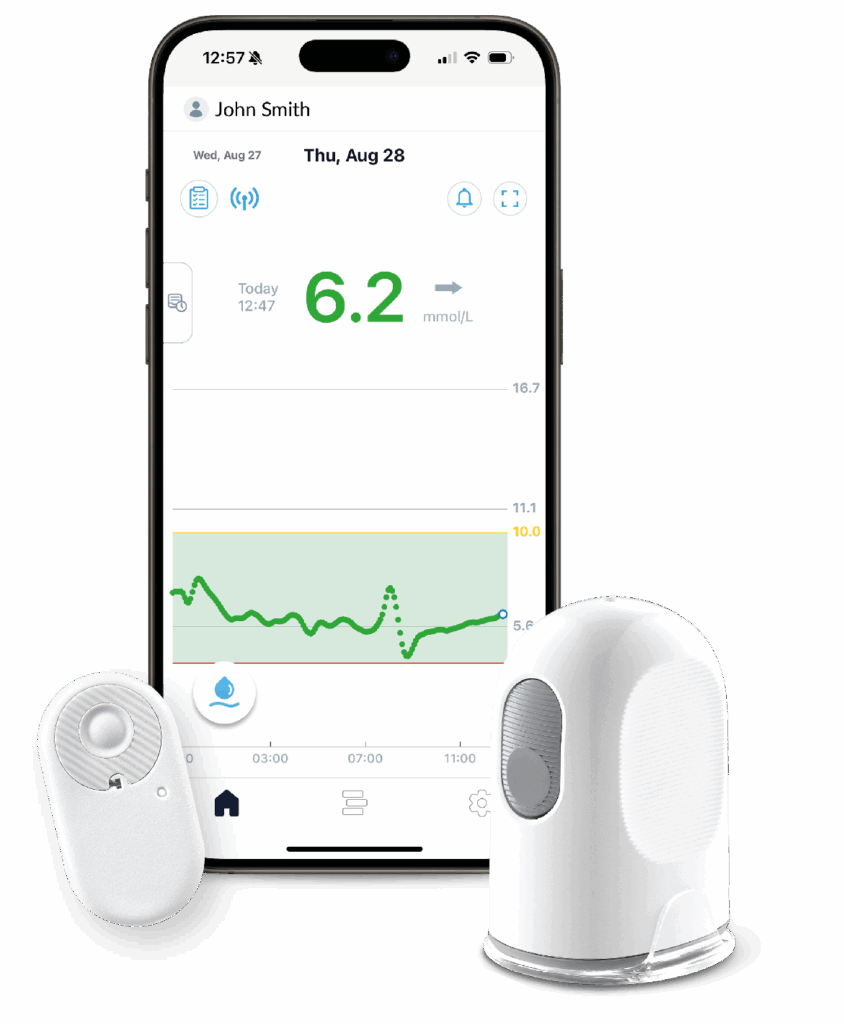
Key Features
Exceptional accuracy¹
MARD 8.7%
Adult 20/20 94%
Adult 40/40 >99.5%
Hyper- & hypo-glycaemic alerts, easily shared with carers *²
Easy to use
device and applicator²
Remotely share data with healthcare provider *
No calibration required, optional
calibration available²
15-day sensor²
30-minute warm-up time²
Optional receiver also
available to reduce
digital inequalities†
*Glucose data sharing with others (carers, HCPs) will not be available until late 2025 when partner Apps are launched.
† Optional receiver currently in development, expected 2026. 1. Jendrike N et al, Journal of Diabetes Science and Technology. 2025; 1–8. 2. 8100-10657 Rev B ALLYcgm User Manual

1
Wash hands and wipe the area where the sensor will be applied
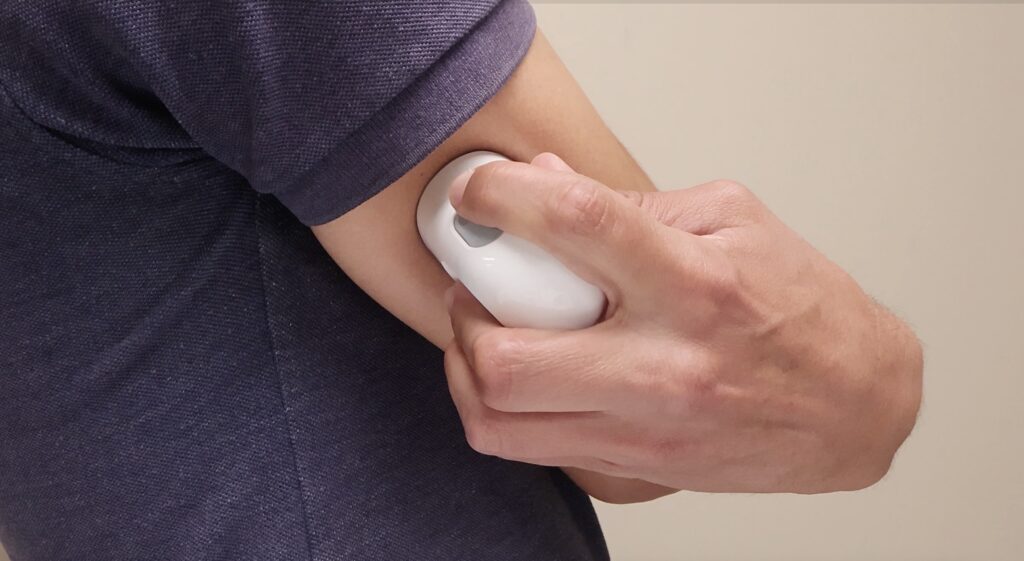
2
Place applicator flat on chosen area and press release button

3
Place applicator flat on chosen area and press release button
Steps shown are a simplified overview; full application steps can be found in the ALLYcgm user manual. 1. 8100-10657 Rev B ALLYcgm User Manual
Easy to use
1
Download ALLYcgm from Apple App Store or Google Play
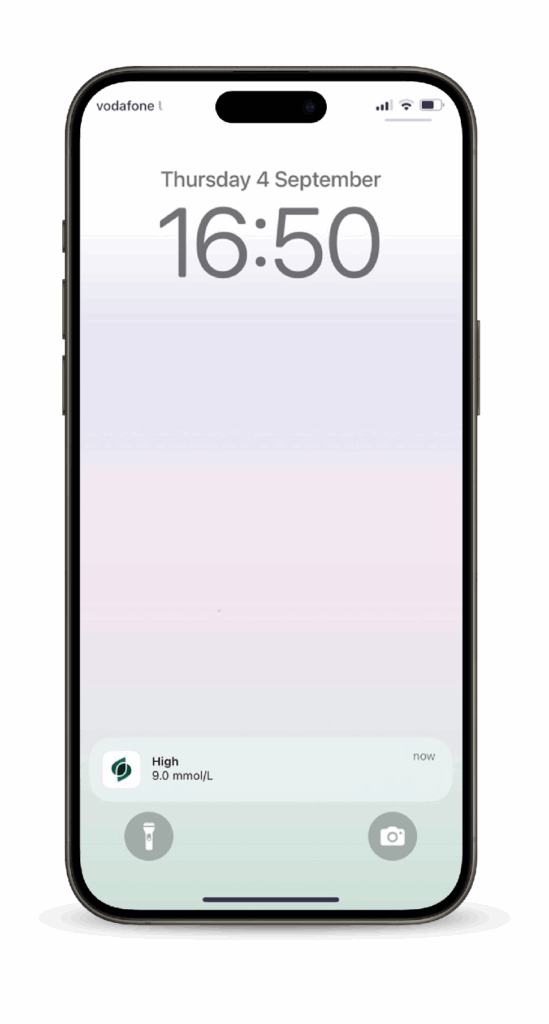

2
Create an account and sign-in
3
Scan the sensor and follow the on-screen instructions

4
Apply the sensor and start your sensor session after the 30-minute warm-up time

Mobile Device sold separately.

Simple data provision for the patient
Patients can view glucose trends, statistics, history and more from the Dashboard.

Images shown are from the ALLYcgm App for sensor users. Mobile device sold separately.

Making accuracy more affordable
Benefits for those living with diabetes.
Benefits for the NHS.
No calibration required¹
Data can be shared with healthcare provider remotely¹
Better value for the NHS*
Easy to use device and applicator¹
15-day sensor with 30-minute warm-up time¹
Alerts for hyper- & hypo-glycaemia which can be shared with carers
*Based on a comparison of the ALLYcgm drug tariff submitted price and the published prices of currently listed CGMs. 1. 8100-10657 Rev B ALLYcgm User Manual. Minimum device and operating system requirements apply. ALLYcgm is indicated for continuous monitoring of blood glucose levels via measurement of glucose in the interstitial fluid in persons with diabetes mellitus aged 18 and older. ALLY® is a registered trademark owned by AgaMatrix, Inc. Apple, the Apple logo and iPhone are trademarks of Apple, Inc., registered in the U.S. and other countries. App Store is a service mark of Apple Inc., registered in the U.S. and other countries. Android is a trademark of Google LLC. Google Play and the Google Play logo are trademarks of Google LLC. ©2025 AgaMatrix, Inc. All rights reserved. 7500-10356 Rev A September 2025.
We use cookies to improve your experience on our site. By using our site, you consent to cookies.
Manage your cookie preferences below:
Essential cookies enable basic functions and are necessary for the proper function of the website.
These cookies are needed for adding comments on this website.
These cookies are used for managing login functionality on this website.
Stripe is a payment processing platform that enables businesses to accept online payments securely and efficiently.
Service URL: stripe.com (opens in a new window)
Statistics cookies collect information anonymously. This information helps us understand how visitors use our website.
Google Analytics is a powerful tool that tracks and analyzes website traffic for informed marketing decisions.
Service URL: policies.google.com (opens in a new window)
SourceBuster is used by WooCommerce for order attribution based on user source.
You can find more information in our Cookie Policy and Privacy Policy.